
What is TPD all about; and why should you care?
Firsty, what do you know about the Tobacco Products Directive?
The Tobacco Products Directive (TPD) or European Tobacco Products Directive (EUTPD) (2014/40/EU) is a directive of the European Union which legislates the sale and merchandising of tobacco and tobacco related products across the EU. The TPD aims to ensure a high level of health protection for European citizens. The Directive entered into force on 19th May 2014 and became applicable in the EU Member States on 20th May 2016.
This Directive covers the manufacture, presentation, and sale of all tobacco related products including cigarettes, roll your own tobacco, and electronic cigarettes. In order to address this situation, the European Union and its Member States have taken various tobacco control measures in the form of legislation, recommendations and information campaigns.
In summary, the policies for the Tobacco Products Directive cover; the regulation of tobacco products on the EU market (e.g. packaging, labelling, and ingredients), advertising restrictions for tobacco products, the creation of smoke-free environments, tax measures and activities against illegal trade, and acquiring anti-smoking campaigns.
With specific regard to e-cigarettes, the TPD introduced new rules which ensure:
-
minimum standards for safety and quality
-
information is provided to consumers of nicotine containing products
-
that protections are put into place to prevent children from starting to use the products
So, what are these new rules?
As of May 2017, any nicotine containing product relating to e-cigarettes/vaping must:
Restrict the maximum volume of e-liquid in a single container to 10ml

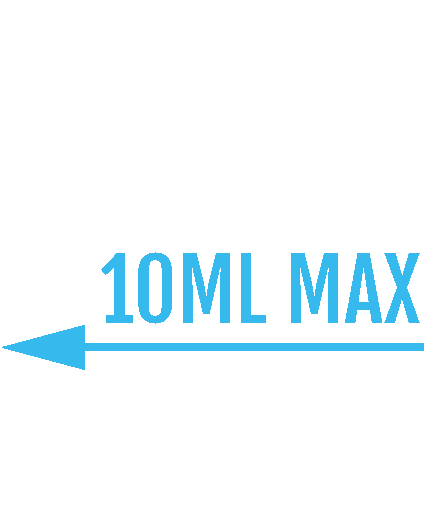
Restrict the capacity of tanks to no more than 2ml
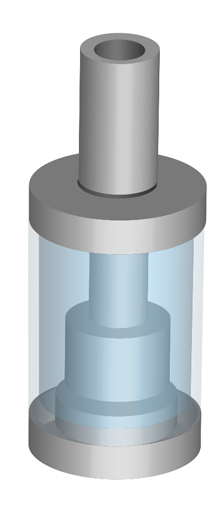

Restrict the nicotine strength of e-liquids to no more than 20mg/ml

Be child resistant and tamper evident

Not contain caffeine, taurine or colourings



Be submitted to the MHRA before they can be sold

Include relevant warnings and information

Between the directive coming into force May 2016, and the cut-off date of May 2017, retailers should have sold any stocks of products that did not comply with TPD.
As of May 2017, you should not be able to purchase non-compliant products within the EU. For this reason, you may have seen certain brands that you were familiar with disappear from the shelves. If this is the case for you, chances are the manufacturer was not able to comply with the new requirements in time.
What has Vape UK been doing?
Since the directive came into action, we have been working to ensure that every product we stock is compliant by the deadline.
For products that we produce ourselves in the UK, such as the popular Brighton Rock range of e-liquids, this involved submitting the products for testing and certification.
We are pleased to say that each flavour passed with flying colours. This is due to the fact that our e-liquids only contain the highest grade ingredients:
- Propylene Glycol (PG)
- Vegetable Glycerine (VG)
- Pharmaceutical grade nicotine
- De-ionised water
- Natural and artificial flavourings
They are also Diacetyl and Acetyl Propionyl free.
What will change at Vape UK?
The changes to the law will affect the products that we can sell. All non-compliant products will be removed from our shelves which means that our e-liquid ranges will reduce.
You will notice that 30ml bottles of e-liquid will be taken off sale and replaced by 10ml bottles. Worry not; we will be re-packaging our products to ensure you still get the same great prices. We want to ensure that our customers do not get hit by unnecessary price hikes as a result of the legal changes.
You may also spot that our tank sizes reduce to a maximum of 2ml. For new and less experienced vapers, the chances are you won't notice the difference. However, for advanced vapers and sub-ohmers, this will likely cause a period of readjustment.
Rest assured that we will continue to stock as wide a range of products as possible to continue catering for all levels of vaper; but you can be confident that everything we sell will be fully compliant with all of the new quality and safety regulations.
Should you have any questions about TPD, or its effect on any of your favourite products, don't hesitate to get in touch.



